n-Butyllithium/N,N,N',N'-Tetramethylethylenediamine-Mediated Ortholithiations of Aryl Oxazolines: Substrate-Dependent Mechanisms | Journal of the American Chemical Society

R)‐BINOL‐6,6'‐bistriflone: Shortened Synthesis, Characterization, and Enantioselective Catalytic Applications - Mouhtady - 2021 - European Journal of Organic Chemistry - Wiley Online Library

ortho-Lithiations Reassessed: the Advantages of Deficiency Catalysis in Hydrocarbon Media | Organometallics
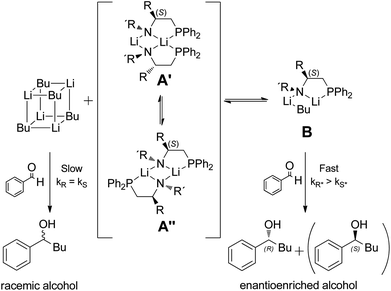
A computational study of the enantioselective addition of n-BuLi to benzaldehyde in the presence of a chiral lithium N,P amide - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C2OB06910E

29 Si CPMAS NMR spectra of (a) carrier C, (b) C treated with BuLi, and... | Download Scientific Diagram
![Concise synthesis of 3-alkylthieno[3,2- b ]thiophenes; building blocks for organic electronic and optoelectronic materials - RSC Advances (RSC Publishing) DOI:10.1039/C9RA08023F Concise synthesis of 3-alkylthieno[3,2- b ]thiophenes; building blocks for organic electronic and optoelectronic materials - RSC Advances (RSC Publishing) DOI:10.1039/C9RA08023F](https://pubs.rsc.org/image/article/2019/RA/c9ra08023f/c9ra08023f-s4_hi-res.gif)
Concise synthesis of 3-alkylthieno[3,2- b ]thiophenes; building blocks for organic electronic and optoelectronic materials - RSC Advances (RSC Publishing) DOI:10.1039/C9RA08023F

NMR and DFT Studies with a Doubly Labelled 15N/6Li S‐Trifluoromethyl Sulfoximine Reveal Why a Directed ortho‐Lithiation Requires an Excess of n‐ BuLi - Hédouin - Angewandte Chemie International Edition - Wiley Online Library




![Solved OZO Br NiBry, Ir[dFppy]z(dtbby)PF6 CsHCO3, BlueLED | Chegg.com Solved OZO Br NiBry, Ir[dFppy]z(dtbby)PF6 CsHCO3, BlueLED | Chegg.com](https://media.cheggcdn.com/media/ed5/ed57a899-fd4a-4095-93ea-6e74405e49f3/phpU6uXgx.png)



![Stats] 2. Bundesliga table after matchday 25. : r/soccer Stats] 2. Bundesliga table after matchday 25. : r/soccer](https://preview.redd.it/7h0vyfk96xl81.jpg?auto=webp&s=ef20eb5c5e005905b30e2903b65f90e8ae894f6c)